The project merges seven European multinational laboratories assembling the interdisciplinary teams from three different countries spanning the fields of different nanosciences including nanochemistry (Poland, Romania), nanophysics (Poland, Romania), molecular nanobiology (Norway, Poland), nanopharmacology (Poland, Norway), nanotoxicology (Norway, Romania), and clinical nanomedicine (Poland). These multinational teams will collaborate together to develop a novel strategy and visionary approaches in the field of nanotechnology-based molecular diagnostics involving mMRI and targeted enzymatic protein/antibody therapies in human lung cancer. A new dimension of this project is to create a unique nanosafety paradigm to address a novel “pharmacovigilance” approach for developing innovative tools used in preclinical risk assessment.
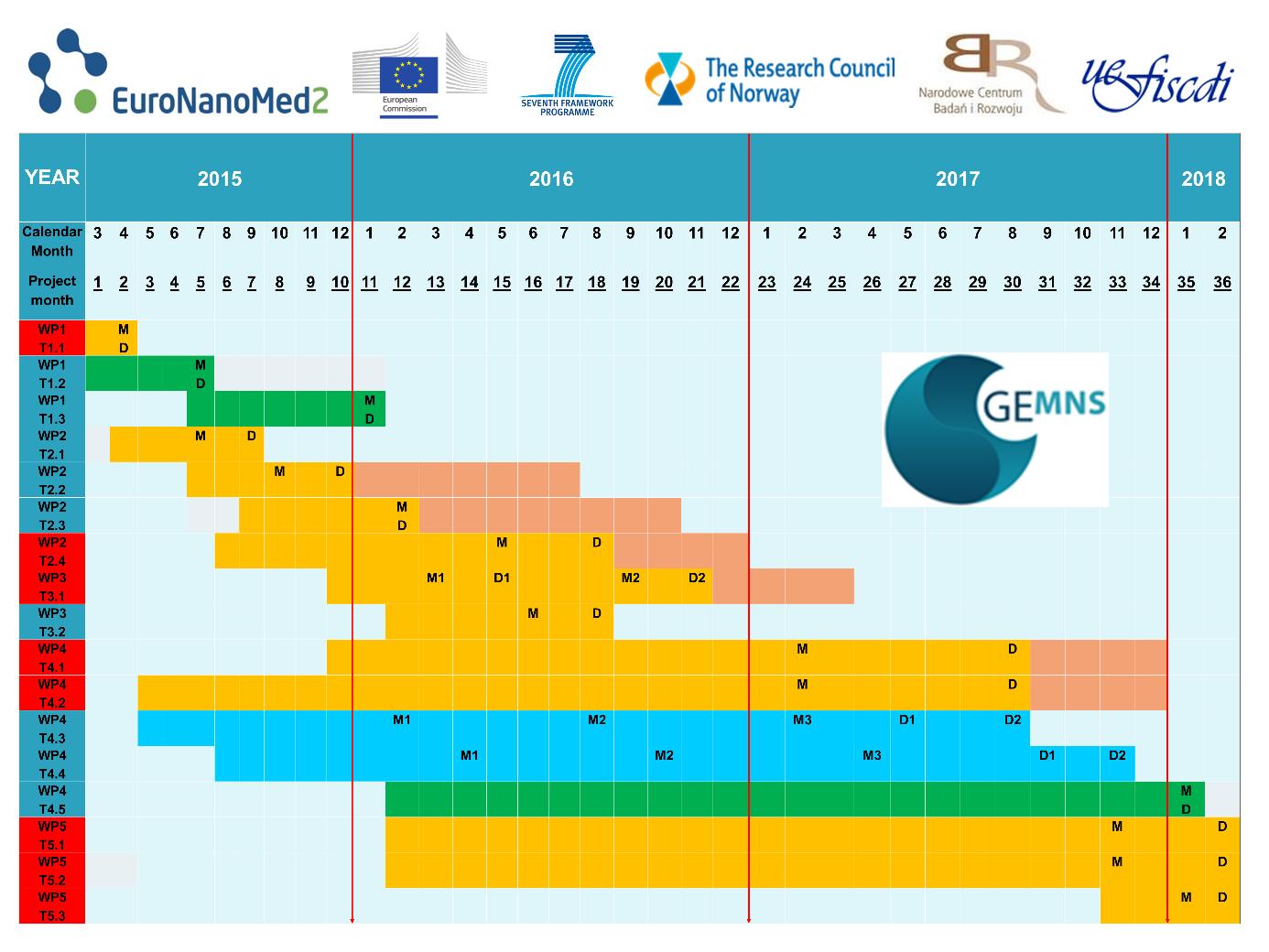
The project is composed of five work packages (WP1, WP2, WP3, WP4 and WP5).
WP1 Molecular modeling and proof of concept
WP2 Chemical synthesis and bio(nano)engineering
WP3 Physicochemical and biological characterizations
WP4 Cellular, molecular and nanosafety in vitro/in silico studies
WP5 Preclinical efficacy in vivo studies and regulatory affair roadmaps
WP1 Molecular modeling and proof of concept
This part of the project is focused on the molecular modeling of self-assembled polymers and enzymatic proteins. The WP1 is designed of three tasks (1.1, 1.2 and 1.3).
Task1.1. Pre-selection of the polymeric nanogels from chemical libraries
Task1.2. Optimalization of polymeric nanogel size to immobilize the enzyme
Task1.3. Modeling of enzyme-polymeric nanogel-GEMNS interactions as a function of temperature
WP2 Chemical synthesis and bio(nano)engineering
This part of the project is focused on the synthesis of graphene-encapsulated magnetic nanoparticles (GEMNS) containing metal or mixture of different metals inside the few layer graphene coat. GEMNS will be modified with polymeric nanogels suitable for absorption of the selected enzymatic proteins. Target monoclonal antibodies will be attached with. The final nano(bio)structure will be treated as a theranostic drug/contrast candidate. The WP2 is designed of four tasks (2.1, 2.2, 2.3 and 2.4).
Task2.1. Synthesis of GEMNS and optimization of their phase composition
Task2.2. Pre-functionalization of GEMNS
Task2.3. Modifications of GEMNS with polymeric nanogels and antibodies
Task2.4. Enzyme loading and thermal-based release enzyme kinetic studies
WP3 Physicochemical and biological characterizations
This part of the project is focused on the comprehensive physicochemical and biological characterization of GEMNS. The WP3 is designed of two tasks (3.1 and 3.2).
Task3.1. Morphological, structural, magnetic, relaxometric and immunochemical characterizations of theranostic GEMNS
Task3.2. Preparation and characterization of stable dispersions of theranostic GEMNS in aqueous solutions
WP4 Cellular, molecular and nanosafety in vitro/in silico studies
This part of the project will answer the fundamental questions in molecular targeting: Did the theranostic GEMNS sufficiently reach its molecular targets? How much of the therapeutic GEMNS and loaded enzyme is in the target cells? Did the amount of the therapeutic enzyme delivered to any cell exceed toxic limits, if any? Is the enzyme co-product of interest is produced to enhance other toxic effects? The WP4 is designed of five tasks (4.1, 4.2, 4.3, 4.4 and 4.5).
Task4.1. Molecular MRI studies with theranostic GEMNS in phantom and cancer cells (in vitro)
Task4.2. Magnetic fluid hyperthermia studies with theranostic GEMNS in phantom and cancer cells
Task4.3. Microfluidic channel approach (lab-on-chip) for nanosafety studies
Task4.4. Cytotoxic, genotoxic and epigenetic screenings (in vitro)
Task4.5. Prediction of theranostic GEMNS toxicities using QSAR strategies (in silico)
WP5 Preclinical efficacy in vivo studies and regulatory affair roadmaps
The central focus of this work package is a thorough understanding of the fundamental principles of the final GEMNS’ behavior in normal and lung cancer cells in vivo to study pharmaco-toxicological outcomes due to X-ray exposures. The WP5 is designed of three tasks (5.1, 5.2 and 5.3).
Task5.1. Diagnostics (MRI) efficacy of theranostic GEMNS in translation animal cancer models
Task5.2. Therapeutics efficacy of theranostic GEMNS in translation animal cancer models
Task5.3. Regulatory roadmaps for translational cancer studies with theranostic GEMNS